Location: Home >> Detail
TOTAL VIEWS
J Sustain Res. 2026;8(1):e260019. https://doi.org/10.20900/jsr20260019
1 Laboratory of Biogeochemistry, Tula State Lev Tolstoy Pedagogical University, Tula 300026, Russia
2 Medical Institute, Tula State University, Tula 300012, Russia
* Correspondence: Leonid V. Perelomov
Contamination of biospheric components with hydrocarbons from oil and petroleum products is one of the most dangerous forms of environmental pollution. This review characterizes new, effective hydrocarbon sorbents—organoclays, demonstrates the complex chemical composition of oil and petroleum products and shows their adsorbate properties based on published data. While existing papers have adequately addressed the adsorption of aromatic hydrocarbons by organoclays, data on alkanes adsorption is practically nonexistent. High efficiency of adsorption and retention of oil hydrocarbons by various organoclays has been demonstrated. Possible models and mechanisms of adsorption of organic substances on organoclays are discussed. Some poorly studied issues concerning the adsorption of oil hydrocarbons by the sorbents under consideration are indicated.
The development of the oil producing and oil refining industry leads to large-scale pollution of water, soils and grounds with oil and oil products [1,2]. Oil spills occur when crude oil leaks during its production, transportation, and storage. These spills can be accidental releases of oil-containing liquids into the environment. Various factors, such as accidents, negligence, and intentional discharge, can lead to oil spills [3]. The main cause of major oil spills are tanker accidents [4] (Figure 1).
Another major cause of oil spills is the daily leakage of fuel and lubricants from cars. There are approximately 1.5 billion vehicles with internal combustion engines worldwide. Therefore, even a small release of oil-containing materials from each vehicle into the environment can lead to a significant environmental problem.
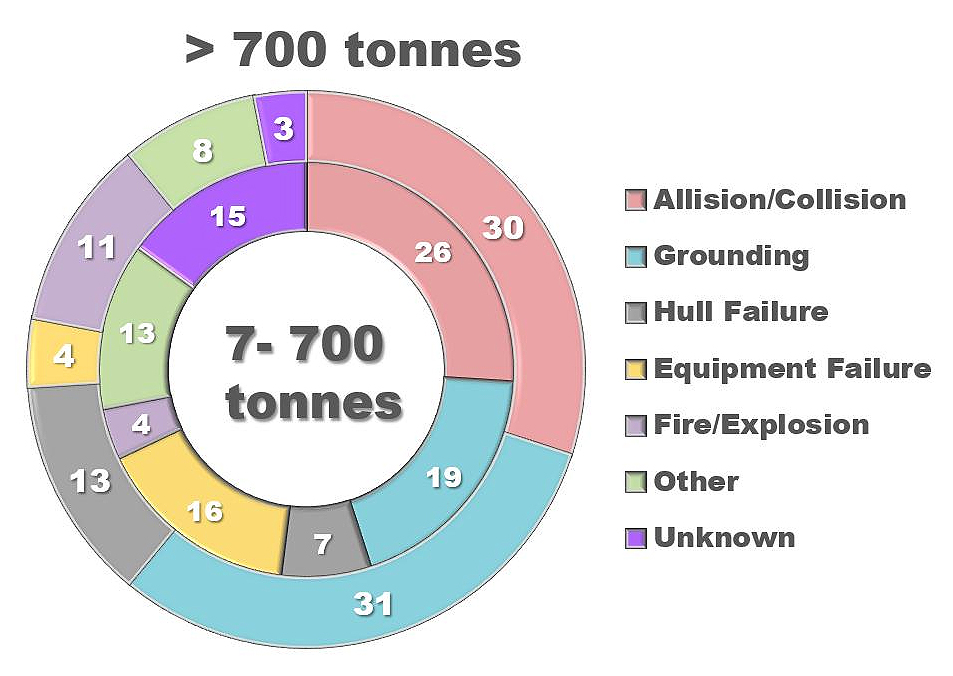 Figure 1. Causes of tankers spills [5]. The inner circle represents the number of smaller oil spills, ranging from 7 to 700 tons, while the outer circle depicts the number of larger spills that are more than 700 tons.
Figure 1. Causes of tankers spills [5]. The inner circle represents the number of smaller oil spills, ranging from 7 to 700 tons, while the outer circle depicts the number of larger spills that are more than 700 tons.
Natural causes of oil spills should also be noted, accounting for approximately 6% of all oil spills. Such spills can occur due to the movement of the Earth’s tectonic plates and the release of oil from layers beneath the seafloor [6].
Another factor leading to oil spills into the environment is its production. During drilling and well preparation, crude oil passes through drilling fluids and various chemical reagents, such as surfactants, acids, salts, and others. During well operation, oil releases into the environment can occur during accidents, startups, and repairs. The main component of these releases is formation fluid, which contains dissolved gas, oil, and formation water. Oil spills into water bodies contribute to the spread of oil and petroleum products over long distances and the contamination of vast territories. Oil spills with similar chemical composition occur during pipeline accidents.
It's worth noting that the number of oil spills from tankers carrying crude oil has decreased significantly over the past fifty years. The number of spills larger than 7 tons has decreased by more than 90% over the past half-century. However, no further decline in the number of tanker spills has been observed recently (Figure 2).
When oil enters the environment, it causes significant damage. The negative effects of pollution can persist for a long time. Living organisms die under the direct influence of oil hydrocarbons or when they alter habitat conditions. Soil degradation and loss of fertility occur. The natural self-purification process is based primarily on the microbial destruction of hydrocarbons and is relatively slow, especially in areas with low temperatures [7].
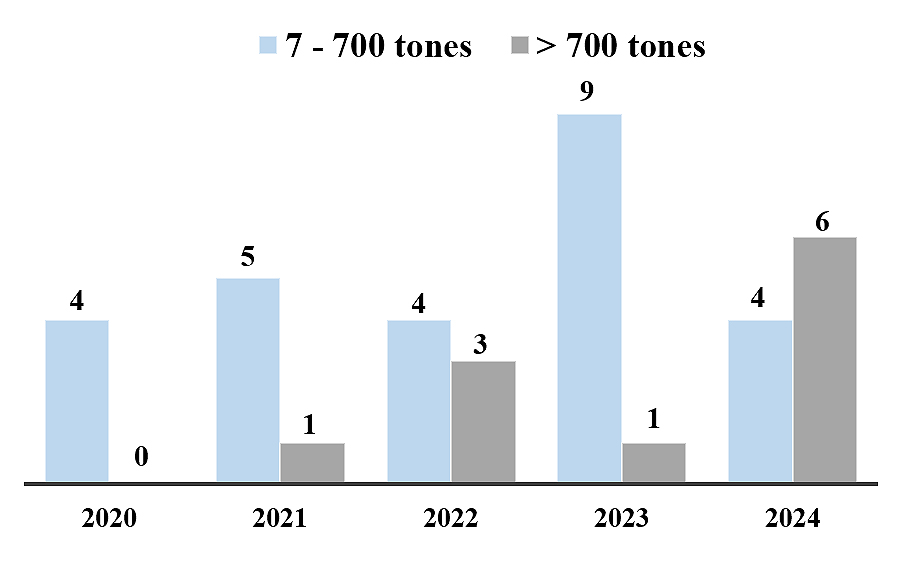 Figure 2. Number of oil spills from tankers since 2020 [5].
Figure 2. Number of oil spills from tankers since 2020 [5].
Thus, the problem of cleaning up the environment from oil spills is one of humanity’s priority tasks within the framework of sustainable development. Addressing this challenge requires developing new and improving existing, proven technologies for restoring oil-contaminated lands and waters.
When selecting a suitable method for the remediation of oil-contaminated ecosystems, several factors must be taken into consideration. These include the nature and composition of the pollutants, as well as the physical, chemical, and biological characteristics of the contaminated area, including the presence or absence of specific microbial communities. Additionally, the time required for remediation, as well as the cost-effectiveness of different technologies, are crucial factors to consider.
Remediation of ecosystems contaminated with oil’s hydrocarbons can be achieved by four main, well-known groups of cleaning method: mechanical, chemical, physicochemical, biological and possible combinations of these methods [8–10].
The main goal of most modern remediation techniques is to isolate, completely or partially remove pollutants, and transform toxic substances into non-toxic or less harmful forms [11]. The effectiveness of cleanup efforts at any contaminated site can be enhanced by integrating various existing technologies. Figure 3 illustrates the advantages and disadvantages of different cleanup methods.
Despite the effectiveness of chemical cleaning in oil-contaminated areas (up to 90%), many of the chemicals used in this process are toxic and have a number of drawbacks [12–14]. Biological treatment is limited due to the long duration required for the remediation process [15]. Physicochemical purification primarily involves the adsorption of oil hydrocarbons onto various adsorbents, with possible subsequent biological methods for decomposition of adsorbed toxicants or removal of adsorption products from the contaminated ecosystem. Adsorption methods involve the use of solid porous substances with a large specific surface area, capable of binding particles of a substance (from the liquid or gas phase) to their surface. Such substances have a wide range of applications in various purification technologies [16]. Natural sorbents such as plant-based materials (peat, peat moss, sawdust, clay, perlite etc.) [17–21] and mineral-based materials like sawdust, clay, and perlite are used for the sorption of oil components. Artificial materials based on viscose, hydrated cellulose, synthetic fibers, and thermoplastic materials are also used [22]. Adsorption methods are the most effective, simple, and safe. Adsorbents used are environmentally stable materials that are readily available and inexpensive [16].
Clay minerals are characterized by unique properties that allow them to be used as adsorbents: they are stable materials, have a high specific surface area and sorption capacity, some clays are capable of swelling. Furthermore, they are readily available, inexpensive, and environmentally friendly. All this determines the use of clay minerals as adsorbents for a wide range of organic and inorganic substances [23]. However, natural clay minerals have a negatively charged surface and are therefore capable of adsorbing mainly hydrophilic substances. Chemical modification of clay surfaces with surfactants, the synthesis of so-called organoclays, makes them hydrophobic. A variety of surfactants, both natural and synthetic, can be used to modify them. Organoclays largely retain the properties of clay minerals, such as a branched surface and stability, but are capable of adsorbing a wide range of hydrophobic organic substances, including oil and its components [24].
Currently, the possibilities of using organoclays in environmental conservation practices are being actively studied based on their structure and properties, sorption capacity and the strength of binding pollutants. Additionally, there is an increase in publications related to the optimization of synthesis methods and the selection of the best modifiers, as well as methods of their application, as evidenced by the increase in the number of publications in the database Sciencedirect by keywords “Organoclays” и “Clay minerals oil sorbents” (Figure 4).
However, there are quite a few works devoted to the use of organoclays for the adsorption of oil hydrocarbons (keywords “Organoclays oil sorbent”) (Figure 4). In our review, we gave a brief description of organoclays as sorbents, showed the complex chemical composition of oil, and attempted to summarize the main data published to date on the patterns of adsorption of oil hydrocarbons by organoclays.
Organoclays: Synthesis and PropertiesThe adsorption capacity of clay minerals is determined mainly by the chemical nature and structure of their pores. However, due to the hydrophilic nature of the surfaces of most natural clay materials, they are usually ineffective in adsorbing organic pollutants such as antibiotics, dyes, pesticides, petroleum hydrocarbons, etc. [25]. With the incorporation of the surfactants in the structure of clay minerals through different chemical and physical processes. The resulting materials combine a large specific surface area, comparable to that of clays, and primarily hydrophobic properties, expanding their application to a wider range of adsorbates, including non-polar organic substances.
Surface-active agents (surfactants) are a group of organic substances whose molecules contain both charged (hydrophilic) and uncharged (hydrophobic) moieties. Surfactants are classified based on products of their dissociation in water: cationic, anionic, amphoteric, nonionic and Gemini surfactants [25].
The production of organoclays is based on chemical reactions (ion exchange, complexation, etc.) and the physicochemical interactions of clay minerals with organic compounds. The most common method for synthesizing organoclays is the reaction of phyllosilicate with a surfactant in an aqueous solution [26]. The presence of water in the interlayer space and surrounding the exchangeable cations amplifies the repulsive forces and leading to an exfoliation of the phyllosilicate sheets offering a total access to their surface area, making easier the adsorption and interaction with surfactants [26]. A number of authors used alcohols as solvents for organic modifiers—ethanol and solutions of ethanol in water [27,28], isopropanol [29], dimethyl sulfoxide (DMSO) and other organic solvents [30]. Sometimes a small amount of acetone is used to synthesize organoclays [31].
Another common method for synthesizing organoclays is the intercalation of organic substances into minerals by fusing phyllosilicates with surfactants. It is generally accepted that the absence of solvents, particularly organic ones, makes organoclay production more environmentally friendly and suitable for large-scale production [32]. The intercalation of organic molecules into the interlayer distance of clays during dry synthesis is confirmed by the expansion of the interlayer distance (d001) using X-ray diffraction [32].
Sometimes a two-step procedure (propping-open procedure) is used to synthesize organoclays. Thus, short chains of fatty acids with a number of less than 10 atoms can be intercalated directly into calcium montmorillonite. But fatty acids with 10–18 carbon atoms are intercalated only after pre-adsorption of hexanol or octanol [33].
When polar organic substances are used as modifiers, they replace water molecules and exchange cations in the interlayer space of smectites and vermiculites [34]. Only cationic and zwitterionic surfactants have a positive charge and therefore have a strong chemical affinity for clay minerals. Their intercalation into phyllosilicate layers occurs through cation exchange, which ensures stable and effective adsorption of these surfactants [35]. Since the number of sorption sites on the surface of clay minerals for binding anionic surfactants is extremely limited, they are adsorbed by minerals much worse than cationic surfactants [36]. Apparently, the primary mechanism of interaction between neutral and non-ionic surfactants and clay surfaces is the formation of complex compounds with interlayer cations. In the case of smectites, adsorption of non-ionic surfactants can be mediated by various chemical interactions: coordination bonds, ion-dipole interactions, hydrogen bonds, acid-base reactions, charge transfer, and van der Waals forces [37].
It is difficult to theoretically estimate the amount of organic substances adsorbed on the organoclay surface based on its composition. Experimental data show that adsorption mechanisms depend on both the type of surfactant molecules and the properties of the adsorbate molecules. Therefore, at present, it is only possible to quantitatively estimate the adsorption of a particular organic substance using experimental methods. Comparison of adsorption isotherms for different organoclays shows that the amount of adsorbed sorbate is determined by the following main structural and physicochemical characteristics:
(1)
(2)
(3)
Since it is difficult to quantify the above three characteristics and numerous combinations of clay mineral, surfactant and adsorbate are possible, it is problematic to construct a theoretical model to estimate the amount of adsorbed substance on organoclay [38].
Oil, Its Chemical Composition and Various Fractions (Petroleum Products)Despite the hydrophobic nature of most petroleum compounds, its components vary in chemical composition and molecular weight. Crude oil is a complex mixture of various hydrocarbons, including paraffins (alkanes), naphthenes, and aromatic compounds, as well as a small amount of organic compounds including sulfur, oxygen, and nitrogen. Furthermore, oil contains small amounts of metal compounds, including vanadium, nickel, and sodium [39]. Olefins are not typically found in crude oil but are formed during the refining process [40]. A distinction is made between paraffinic, naphthenic, and mixed crude oils, depending on the predominance of paraffinic, aromatic, naphthenic, and asphaltene hydrocarbons. Mixed crude oils contain comparable amounts of paraffinic and naphthenic-aromatic hydrocarbons [41]. Low-carbon, high-hydrogen and high American Petroleum Institute (API) gravity crude oils are typically rich in paraffins and tend to produce more gasoline and light petroleum products; high-carbon, low-hydrogen, low API gravity crude oils are typically rich in aromatics [39]. Crude oil can contain up to several thousand different hydrocarbon compounds (Figure 5).
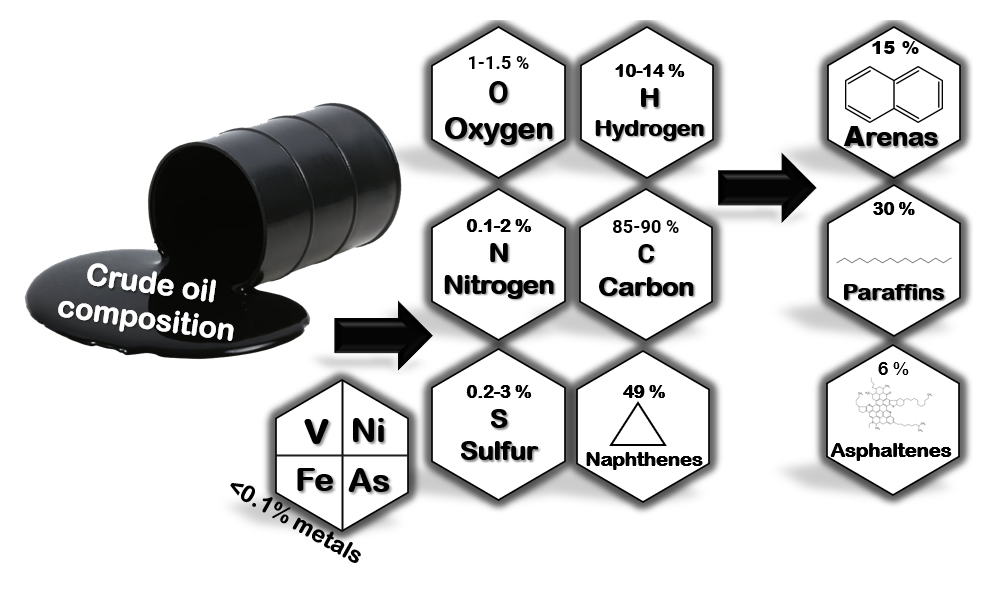 Figure 5. Components of crude oil [42].
Figure 5. Components of crude oil [42].
Since a virtually infinite number of different hydrocarbon mixtures are possible, there are a vast number of types of crude oil. The main groups of hydrocarbons found in oil are:
Paraffins (Alkanes)Alkanes, or saturated hydrocarbons, are the main components of crude oil and most petroleum products. The carbon atoms that make up the carbon backbone are linked together to form a chain (linear or branched alkanes), a ring (cyclic alkenes), or a mixture of both. Linear and branched alkanes are called paraffins. These compounds have an empirical formula of CnH2n+2. Linear alkanes, i.e., alkanes whose carbon atoms are arranged in a straight chain or row, are called normal paraffins (normal alkanes). The branched alkanes are often called isoparaffins (isoalkanes) [43]. The majority of petroleum paraffins have a carbon chain of 17–35 atoms. Some paraffins are liquids and have a melting point of ≤27 °C.
NaphthenesNaphthenes (cycloalkanes) are saturated hydrocarbons that have at least one ring of carbon atoms. They have the general formula CnH2n. The density and boiling point of naphthenes are higher than those of alkanes with the same number of carbon atoms. Naphthenes, commonly found in crude oil, are rings with five or six carbon atoms. These rings often have alkyl substituents attached to them. Polycyclic naphthenes are present in the heavier fractions of crude oil. Examples of nаphthenes include cyclopentane, cyclohexane, methylcyclohexane etc. [44].
AromaticsAnother major type of petroleum hydrocarbons are aromatic compounds (arenes). Aromatic hydrocarbons typically have the empirical formula CnH2n−6 and contain a six-membered carbon ring. The ring has three carbon-carbon double bonds. This arrangement of three double bonds within a six-membered ring imparts special chemical properties of these compounds. Benzene, and all the larger arenes, has a characteristic planar structure forced on them by the electronic requirements of the six (or more) π electrons. The aromatic hydrocarbon group contains not only benzene but also derivatives such as toluene (C6H5CH3), the isomeric dimethyl benzene derivatives (CH3C6H4CH3) etc. [45]. The higher aromatics such as naphthalene and benzo(a)pyrene contain more than one benzene ring. The benzene rings are fused where each ring shares two carbon atoms with its neighboring ring. These latter two compounds are part of a class of hydrocarbons called polynuclear aromatic hydrocarbons (PAHs) [43].
OlefinsOlefins (alkenes or unsaturated hydrocarbons) are hydrocarbon compounds with the general formula CnH2n containing at least one carbon-to-carbon double-bond. Olefins are generally thought to be produced in refineries and petrochemical plants and are not natural components of crude oil.
One of them is associated with the migration-contamination process, in which light oils act as solvents for syndepositional olefins formed along the migration path or within the reservoir section. The other two sources for olefins in oil involve abiogenic alteration of the crude after it has entered the trap. “Cold” radiolytic dehydrogenation of saturated petroleum hydrocarbons, formed as a by-product of the decay of radioactive elements (uranium, thorium, etc.) in the minerals of the formation, is possible. Pyrolysis is also possible, driven by the thermal action of igneous intrusions located near oil reservoirs [46].
In addition to these compounds, crude oil contains a number of surface-active compounds as impurities. They are found in a wide range of petroleum fractions, but are more common in heavier fractions such as resins and asphaltenes. They are considered to be polar components that contain oxygen, nitrogen, and sulfur with very strong interfacial activity that can alter the wettability of mineral surfaces [47].
Crude oil is a complex mixture of organic substances that can be separated by fractional distillation. Therare more than 2000 known types of petroleum products that are obtained from crude oil. The main petroleum products of interest for this review are listed in order of increasing molecular weight from the bottom to the top in Figure 6 [48].
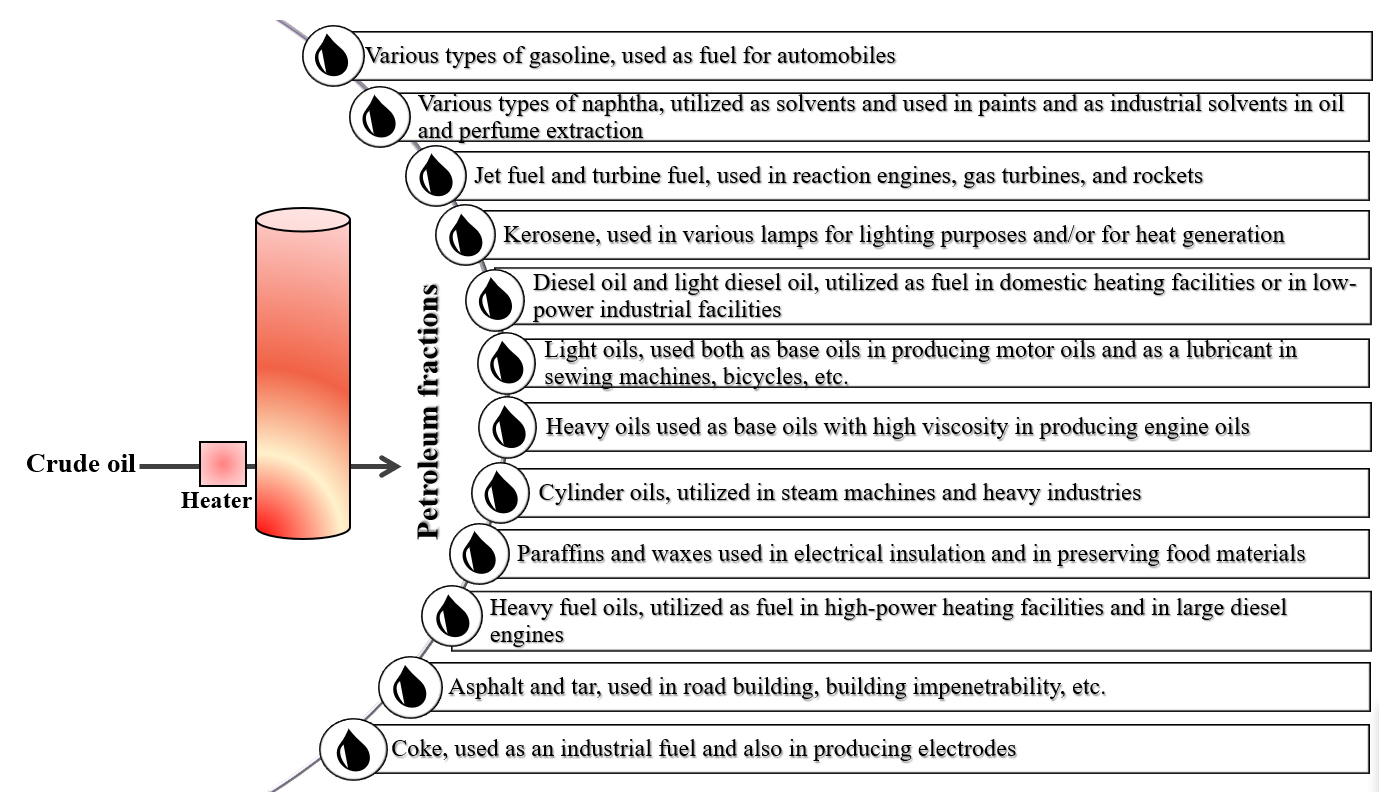 Figure 6. The main petroleum products in order of increasing molecular weight [48].
Figure 6. The main petroleum products in order of increasing molecular weight [48].
A large number of studies have been devoted to the absorption of crude oil by organoclays, mainly based on cationic surfactants. So, the data of authors showed that organoclay prepared by exchanging of the hexadecyltrimethylammonium bromide (HDTMA-Br) with sodium ions in montmorillonite structure sorbed (hold) more crude oil, kerosene, gasoline and toluene than untreated clay. Also, the organoclay sorbed more gasoline and toluene than kerosene and crude oil. For organoclays sorption capacity determination, a method based on ASTM F726-99 [49] was used. This method is based on determining the efficiency of sorbents in absorbing (retaining) non-emulsified oils and other water-immiscible liquids, as well as obtaining laboratory data that can be used to compare the sorption capacity of one adsorbent with another and, thus, their relative cost-effectiveness. In fact, this method provides information on the retention of oil and petroleum products by porous materials, which depends on the viscosity of the substance, rather than on the classical physicochemical adsorption of substances by adsorbents. In this regard, experimental results showed that organoclay adsorbed (retained) hydrocarbons in quantities exceeding its own weight by 5 times [50].
Laterite- and bentonite-based organoclays with two surfactants, hexadecyltrimethylammonium chloride (HDTMA-Cl) and tetradecyltrimethylammonium bromide (TDTMA-Br) were effective in sorbing diesel and engine oil from aqueous emulsions. Montmorillonite-based organoclays had a higher adsorption capacity, with HDTMA-Cl being the most effective modifier [51]. To determine the sorption capacity (retention capacity), a similar method was used, which involves contact of the adsorbent with the oils, followed by draining off the excess and weighing [52].
Commercial organoclay on the base of bentonite was used to remove oil from water. As oil in the oil-in-water emulsions used standard mineral oil, two cutting oils, refinery effluent, and produced water from production wells at Estevan, Saskatchewan. The concentrations of oil in oily waters varied from 26 to 381 mg/L. Batch studies showed that the equilibrium time for the sorption of oil by organoclay was less than 1 h for all emulsions. Adsorption experiments showed that Freundlich model is most appropriate isotherm for standard mineral oil, one of cutting oil and produced water. The Langmuir and BET isotherms fitted well the sorption data for the second cutting oil and refinery effluent, respectively. The results indicated that the organoclay can remove up to 100% oil from oil-in-water emulsions. Organoclay was an effective medium for treating oily waters [53].
The use of organoclays based on Wyoming Na-Montmorillonite with three cationic surfactants: octadecyltrimethylammonium bromide (ODTMA-Br, C21H46NBr), didecyldimethylammonium bromide (DDDMA-Br, C22H48BrN), and ROKAmin SR5 (hydrogenated tallow), dimethylammonium chloride (tallow) for the adsorption of diesel, hydraulic oil and engine oil has been shown the greatest efficiency for the sorption of diesel of the third adsorbent, for the sorption of hydraulic and engine oil—organoclay based on DDDMA-Br. The sorption capacity of organoclays in relation to hydrocarbons depended on the surfactants used in the synthesis of them. Two long carbon chains in molecules of DDDMA-Br and tallow appear to enhance the sorption capacities of the organoclays with their participation. In contrast, the mineral modified with ODTMA-Br sorbed significantly less hydrocarbons [17].
Organoclays synthesized with the participation of cationic surfactants—quaternary ammonium salts: distearyldimethylammonium chloride (DSDMA-Cl) and alkyl dimethyl benzyl ammonium chloride (ADMBA-Cl), effectively absorbed gasoline, kerosene and diesel fuel. The study of the swelling of the organoclay according to Foster shows that all organoclays exhibit better sorption capacity in all oil products compared to unmodified clay. Modification of clays by DSDMA-Cl resulted in the formation of organoclays with better sorption properties in relation to the studied petroleum products compared to the ADMBA-Cl modification. The use of a surfactant mixture (DSDMA-Cl/ADMBACl) for modification showed slightly higher efficiency compared to modification with individual surfactants (DSDMA-Cl).
For DSDMA-Cl synthesized organoclays, the adsorption capacity follows in descending order: diesel fuel > kerosene > gasoline. The best result was obtained with diesel fuel, which probably indicates a better interaction of organoclay with this oil product. This interaction is directly related to the chemical composition of the compounds (gasoline: C5–C10; kerosene: C11–C12; diesel: C13–C17). The results may also be related to the solvent viscosity, considering that diesel has the highest viscosity. In the case of organoclays with ADMBACl, the order is the following: gasoline ≈ diesel > kerosene.
The study of the basal reflection value of the obtained organoclays by X-ray diffraction showed that the basal distance (d001) of the organoclays increased after treatment with quaternary ammonium salts, which confirms the intercalation of ammonium cations into the clay. The largest basal distance was observed in the clays treated with ADMBA-Cl surfactant. Infrared spectra revealed new bands of CH2 and CH3 vibrations corresponding to the presence of ammonium salts in the clay structure [54].
Bentonite modified with cetyltrimetylammoniumbromide (CTA-Br), a quaternary ammonium compound which possesses surfactant properties was tested as adsorbent for petrol, kerosene and automotive gas oil. The most effective was the use of organoclay in relation to automotive gas oil the adsorption of kerosene and gasoline was approximately the same. Increasing the dose of the modifier for the synthesis of organoclays somewhat reduced the adsorption of oil products [55].
Despite the relevance of the problem, the adsorption of oil paraffins by organoclays has not been studied enough. Low-molecular alkanes are often used as solvents in studying the adsorption of aromatic hydrocarbons [56], and often have a significant effect on the sorption process. However, the adsorption of oil paraffins themselves has been practically not studied. Ref. [26] notes that the use of surfactants with long alkyl chains, such as hexadecyltrimethylammonium (HDTMA), in the synthesis of organoclays leads to the formation of sorbents that effectively adsorb alkanes or hydrophobic compounds. Modifiers with aromatic rings, such as benzyldimethyltridecylammonium (BDTA) or benzyltrimethylammonium (BTA), demonstrate excellent affinity for aromatic compounds (Figure 7).
The use of organoclay obtained on the basis of sodium montmorillonite and cationic surfactant dimethyldioctadecylammonium chloride (DODMA-Cl) as an adsorbent for oil containing 67 wt% paraffinic compounds and 33 wt% naphthenic compounds (Marcol 52, Exxon Mobil) resulted in more efficient adsorption of paraffin oil compared to montmorillonite. Multiple regeneration of the sorbent by washing with N,N-dimethylcyclohexylamine preserved the sorption capacity of the organoclay and even increased it [57]. The authors explain this phenomenon by the hydrophobic nature of the adsorption surface and the large interlayer distance in the organoclay.
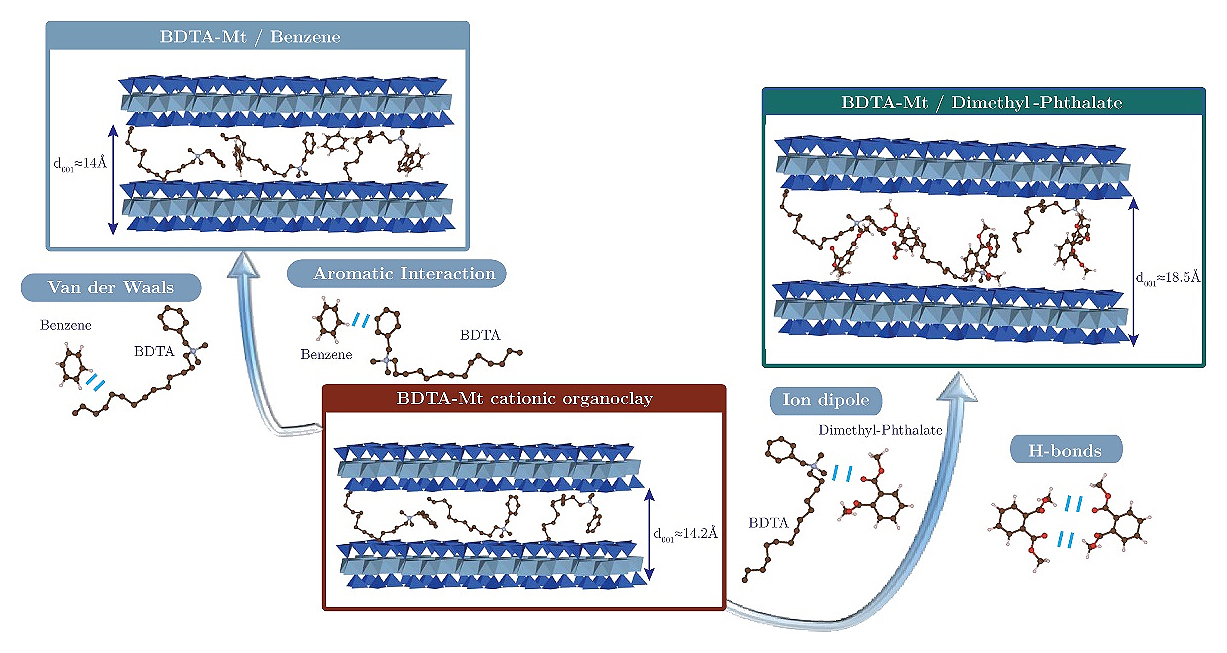 Figure 7. Possible adsorption mechanisms in a BDTA-Mt cationic organoclay showing a lateral surfactant arrangement involved in the sequestration of benzene, a nonpolar hydrophobic organic compound [26].
Figure 7. Possible adsorption mechanisms in a BDTA-Mt cationic organoclay showing a lateral surfactant arrangement involved in the sequestration of benzene, a nonpolar hydrophobic organic compound [26].
There is also little data on the adsorption of naphthenes by organoclays. The study of the adsorption of volatile compounds-cyclohexane (naphthene) together with aromatic hydrocarbons-toluene and chlorobenzene on natural, sodium and didodecyldimethyl ammonium bromide-modified phyllosilicate (mixed-layer illite/smectite) by the chromatographic method showed that the adsorption capacity of organoclay with respect to cyclohexane reaches 2.5 mg/g. At the same time, adsorption capacity for toluene, and chlorobenzene were 2 and 3.5 mg/g respectively. The hydrocarbons adsorption isotherms were of type II of the BET classification therefore the sorption was multi-layered. The isotherms showed that the intercalation of didodecyldimethyl ammonium bromide (double-chain cationic surfactant) significantly improved the naphthen and aromatic organics adsorption. In the case of a mixture of cyclohexane and chlorobenzene, the adsorption isotherms also belonged to type II according to the BET classification. At the same time, the amount of both substances adsorbed by the organoclay decreased, which may indicate their competition for sorption sites, despite differences in hydrophobicity [56].
The adsorption of aromatic substances contained in oil has been studied quite well using a variety of organic substances as an example. Hexadecyltrimethylammonium bromide (HDTMA-Br) modified (surfactant with long alkyl chain) montmorillonite and commercial organoclay PM199 were used to investigate naphthalene and gasoline adsorption. An experimental study showed that HDTMA-Br organoclay adsorbed 3.5% (by weight) of naphthalene and 6.9% of gasoline; commercial organoclay PM199 adsorbed 21.8% of gasoline from a 200 μg/mL solution using a methanol/water mixture as the solvent. Sorption of naphthalene and gasoline hydrocarbons by organoclays is due to short-range interaction forces between sorbates and organophilic sorbents. The interlayer expansion of organoclay with HDTMA-Br upon interaction with naphthalene solution by approximately 2 Å (19.44 Å–21.32 Å) clearly indicates that naphthalene molecules penetrate into the interlayers of organoclay. The basal distance in organoclay with HDTMA-Br upon gasoline adsorption reached only 20.1 Å. The large size of the alkane molecules in gasoline (up to 10.5 Å) resulted in less adsorption of gasoline hydrocarbons and a less pronounced interlayer expansion (0.66 Å). Organoclay PM199, with an initial interplanar spacing (determined by the basal reflection d001 by XRD) of 35.58 Å, was more effective at adsorbing organic compounds with larger molecular sizes. During the sorption of gasoline hydrocarbons, sorbate molecules were able to penetrate the interplanar space and expand it by 3.0 Å to 38.66 Å [58]. For organoclays with long carbon chain of modifier partition has been regarded as the predominant sorption mechanism. The surfactant aggregates on these organoclays can form nano-sized organic phases, which are the main accommodation place for the sorption of organics [59].
In our studies, organoclays based on amphoteric surfactants (disodium cocoamphodiacetate and sodium cocoiminodipropionate) and nonionic surfactants (lauramine oxide, cocamide diethanolamine, and alkyl polyglucosides) were used to adsorb PAHs—high-molecular-weight benzopyrene and low-molecular-weight naphthalene. The results showed that the adsorption capacity of organoclays for PAHs increased compared to the original bentonite, with benzoаpyrene adsorption being higher than naphthalene. The benzopyrene adsorption process by the studied adsorbents was best described by the Langmuir isotherm (R2 = 0.77–0.98). In contrast, naphthalene adsorption was better described by the Freundlich equation (R2 = 0.86–0.96), which may indicate its multilayered nature. It was found that organoclays synthesized with disodium cocoamphodiacetate were most effective at adsorbing benzopyrene, while those synthesized using sodium cocoaminodipropionate were more effective at adsorbing naphthalene [24].
Organoclays with small, compact organic cations of modifier, such as tetramethylammonium (TMA) can take up hydrocarbons without undergoing appreciable interlayer expansion [60]. The sorption of three aromatic molecules by TMA modified montmorillonite was found [61,62] to decrease in the order benzene > toluene > xylene which was inversely related to molecular size.
Bentonite clay organophilizated by the intercalation of quaternary ammonium salts such as cetylpyridinium chloride and benzalkonium chloride were used to study the removal percentage of benzene, toluene and xylene in gasoline. Initial and final concentrations of benzene, toluene and xylene compounds were determined by headspace gas chromatography. 0.216 (60.3%), 0.346 (60.1%) and 0.125 (50.4%) mg/g of benzene, toluene and xylene respectively were removal by 1 gram of organoclay with cetylpyridinium chloride from 15% (volume) emulsion of gasoline in water [63]. Organoclay with cetylpyridinium chloride showed the largest sorption capacity for gasoline in accordance with [64,65]. The sorption value was 3.7 times higher (about 4.4 g/g) when compared to the natural mineral. The affinity order for this organoclay was: gasoline > diesel oil, while for organoclay with benzalkonium chloride is reversed (diesel oil > gasoline) [63].
Organoclays based on montmorillonite and didecyldimethylammonium bromide did not promote the biodegradation of crude oil polycyclic aromatic compounds, especially low-molecular ones, such as dimethylnaphthalenes. This may serve as indirect evidence for a stronger bond between arenes by organoclays compared to the original mineral, the adsorption of PAHs on which promoted their more efficient biodegradation and retained their ability to volatilize. Organoclay based on saponite and didecyldimethylammonium bromide also did not enhance the microbial degradation of dimethylnaphthalenes, but enhanced the biodegradation of some other PAHs, such as phenanthrene [66].
Organoclay with long-chain alkyl quaternary ammonium cation, hexadecyltrimethylammonium bromide (HDTMA-Br) was used to sorb phenol, which is present in small quantities in petroleum but is more common in distillates, and its derivatives from water emulsion. The results of the study showed that the sorption of phenols by this organoclay increases with increasing molecular hydrophilicity (with the addition of chlorine): phenol < p-chlorophenol < 2,4-dichlorophenol. The mechanisms by which these organic substances are adsorbed by organoclay include both adsorption and distribution, with adsorption being the dominant mechanism at low concentrations and distribution at high concentrations [67].
Efficient adsorption of toluene on bentonite modified with cetyltrimethylammonium (CTMA+) is shown in the work [68]. Organoclays used in permeable barriers significantly reduced concentrations of fluorene, pyrene, anthracene, naphthalene and other organics in wastewater [69].
Based on existing laboratory studies, organoclays are successfully used in environmental cleaning practices. For example, an oil platform in the Gulf of Mexico spilled approximately 3 million barrels of oil. Organoclay producing company supplied special organoclay to an environmental remediation contractor working on the oil spill cleanup. The contractor initially conducted laboratory tests and then developed a treatment system using the organoclay. The treatment system effectively treated millions of gallons of contaminated water [70].
In the United States, a pipeline rupture resulted in a large spill of oil into a nearby river. An organoclay producing company was hired to assist with surface water cleanup in the stream. Twelve large hydraulic fracturing tanks were filled with contaminated water from the crude oil spill. After using a dissolved air flotation system, the contaminated water flowed into pressure vessels containing organoclay, which absorbed 75% of its own weight in oil. After three months of treating nearly fifty-five million gallons of water, the organoclay-based system reduced high concentrations of oils and petrochemical hydrocarbons, achieving a 99% reduction. The spent organoclay was disposed of in accordance with state and federal regulations [71].
Organoclays are used to create permeable reactive barriers to remove non-aqueous liquid phases and poorly soluble organic matter from groundwater. Organoclay pre-filters can also be installed before activated carbon filters [72]. A wastewater treatment system at an Air Force base in Utah, USA, includes flocculation, air flotation, organoclay granule columns, an air stripper, and activated carbon columns. The organoclay, a lard-treated dimethyl dehydrogenated montmorillonite, is contained in three tanks operating at a flow rate of 350 gallons per minute. The organoclay in these columns is mixed with anthracite, which acts as a filter material and minimizes the problem of organoclay swelling. This system operated at a 30% lower cost thanks to the use of organoclay filters [73]. Organoclays have been successfully used to purify well fluids from offshore platforms to produce water suitable for discharge overboard. These well waters were acidic and had inlet oil concentrations of 50 to 800 ppm. Initial purification was performed using fixed-bed columns of 30% organoclay and 70% anthracite. Later, radial-flow containers containing 100% organoclay granules were used with great success. This technology has also been used to purify produced water that accompanies oil production at the wellhead [74]. An organoclay-based oil-contaminated water treatment and recycling system was developed and installed at an air conditioning and heating components manufacturing facility in the Southeastern United States. The use of organoclays resulted in higher water clarity and more effective oil and grease removal compared to alum treatment. Organoclay columns cost $0.50 per 10,000 gallons of purified water at an average inlet oil and grease concentration of 25–30 ppm. Under this operating mode, the filter organoclays in the columns require replacement approximately every 3–4 months [73].
Despite the development of alternative energy sources, oil continues to be an important source of energy and raw material for producing automobile fuel and petrochemicals. However, the large-scale production and refinement of oil can lead to significant environmental pollution at every stage of the oil and petroleum product cycle. To combat this issue, modern methods such as the use of modified clay minerals with surfactant additives, known as organoclays, have been developed. These organoclays offer a range of benefits, including increased interlayer spacing and hydrophobic properties, making them ideal for use as sorbents to remove oil hydrocarbons from contaminated environments. The use of different types of surfactants during the modification process allows for the creation of customized clay-based materials with tailored properties, further enhancing their effectiveness in oil spill clean-up operations.
Numerous studies have demonstrated the effectiveness of organoclays for the adsorption of crude oil and its various distillates, such as gasoline, kerosene, and lubricants. However, very few studies have examined the adsorption of individual chemical compounds found in crude oil, particularly paraffins and naphthenes. The adsorption of aromatic hydrocarbons found in crude oil and petroleum products, including benzene, naphthalene, toluene, phenol, polycyclic aromatic hydrocarbons (PAHs), etc., has been studied in more detail and demonstrates the high efficiency of organoclays.
Traditionally, most studies don’t really focus on oil adsorption by sorbents. Instead, they concentrate on oil retention, which can be several times the mass of the sorbent. There are very few studies that actually look at the adsorption of individual hydrocarbons on the surface of organoclays. The concentration of these hydrocarbons is determined using chromatographic methods.
The issue of competition for sorption sites between different groups of oil hydrocarbons and between various hydrocarbons within a group is an interesting and poorly understood area. Knowing the adsorption specificity would help to solve problems related to cleaning natural environments and separating hydrocarbons of different groups in production.
Furthermore, potential pathways for organoclay regeneration, ecotoxicity of waste materials, and methods for their safe disposal are relevant areas for future research on this topic.
Thus, organoclays, based on clay minerals (mainly the smectite group) and various surfactants, are effective adsorbents and retainers of oil, its distillates, and components, and can be used for the remediation of contaminated environments. However, there are some negative aspects to using organoclays in oil spill response, such as cost, biodegradability, and the potential for recycling.
All data generated from the study are available in the manuscript.
Conceptualization: LVP, IVP; Methodology: LVP, MMG; Formal analysis and investigation: LVP, MMG; Writing—original draft preparation: MMG, LVP; Writing—review and editing: LVP, MMG, IVP; Funding acquisition: LVP; Supervision: LVP.
The authors have no relevant financial or non-financial interests to disclose.
The article was prepared as part of the state assignment of the Ministry of Education of Russia on the topic “Synthesis and properties of organoclays for the adsorption of oil hydrocarbons” (Agreement No. 073-00065-25-01 dated 03/18/2025).
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
23.
24.
25.
26.
27.
28.
29.
30.
31.
32.
33.
34.
35.
36.
37.
38.
39.
40.
41.
42.
43.
44.
45.
46.
47.
48.
49.
50.
51.
52.
53.
54.
55.
56.
57.
58.
59.
60.
61.
62.
63.
64.
65.
66.
67.
68.
69.
70.
71.
72.
73.
74.
Perelomov LV, Gertsen MM, Perelomova IV. Organoclays are effective sorbents for hydrocarbons from crude oil and petroleum products. J Sustain Res. 2026;8(1):e260019. https://doi.org/10.20900/jsr20260019.

Copyright © Hapres Co., Ltd. Privacy Policy | Terms and Conditions